This article has since been published in the University of Sheffield’s Biology magazine.
“Over the oxygen supply of the body carbon dioxide spreads its protecting wings.” – Friedrich Miescher, Swiss physician, 1885
Carbon dioxide is usually seen only as a waste gas, while lactate is often treated as a simple by-product of exertion during exercise, and nothing more. In reality, the balance between the two may reveal something deeper about whether metabolism is efficient, oxidative, and protective, or stressed, inflammatory, and degenerative.
CO2 vs Lactate
Ordinarily, carbon dioxide (and bicarbonate) is typically thought of with respect to the regulation of pH, and only in a very general sense, but its essential and protective roles extend far beyond this. Because carbon dioxide plays an important role in keeping the pH of the blood within a narrow range, it has been commonly thought of as a toxin, since a supraphysiological excess can cause unconsciousness and acidosis. Increasing carbon dioxide, however, doesn’t necessarily cause acidosis, and acidosis caused by carbon dioxide isn’t nearly as harmful as lactic acidosis – the accumulation of too much lactic acid in the body, often due to a lack of carbon dioxide (Kamel et al., 2019).
Carbon dioxide and lactate appear to have an inverse relationship, most likely explained by the “Randle cycle”. In 1963, the British physiologist Sir Philip Randle, researching at Oxford University at the time, observed that glucose and fatty acids compete for oxidation in the cell (Randle et al., 1963). He realised that the mitochondrion can only oxidise either glucose or fatty acids at any given time, not both. Therefore, if an influx of fatty acids are present in the bloodstream, whether that be due to stress, inflammation, or just dietary intake, glucose gets “outcompeted” for oxidation in the mitochondria. As a result, much of the remaining glucose can only be burnt through glycolysis, a less efficient process which occurs outside of the mitochondria, rather than through efficient oxidative phosphorylation in the mitochondria. Glycolysis produces no carbon dioxide, rather producing lactate instead. The complete metabolism of glucose, critically involving oxidative phosphorylation, produces more carbon dioxide (and no lactate) per molecule of oxygen compared to glycolysis, or the oxidation of fatty acids.
This is important. This is because carbon dioxide is protective, since it begets effective metabolism, and thus better structure and function and so on. More carbon dioxide in our blood leads to a more efficient usage of oxygen, through the Bohr effect, and an improved blood supply to the tissues due to its function as a vasodilator.
A reduction of carbon dioxide however, caused by hyperventilation for example, can provoke seizure, cause muscle spasms (due to an inability to relax the muscle, which is a high-energy requiring state), promote vascular leakiness, and contribute towards inflammation and potentially even thrombosis events (Salvati and Beenhakker, 2019; Lewis, 2025; Nagy et al., 1985, Curley et al., 2010; Dai et al., 2006; Staubli et al., 1994). Small fluctuations in serum oxygen and carbon dioxide levels occur many times throughout the day, but chronically reduced carbon dioxide – or chronically elevated lactate – can have more serious consequences.
Acute lactate rises (like from exercise) aren’t necessarily concerning, but chronically elevated lactate seen in hypoxia and poor metabolic states leads to pro-inflammatory signalling, cell proliferation (and thus tumour growth), fibrosis, and cardiolipin disruption (Kawauchi, et al., 2008; Liu et al., 2023; Longhitano et al., 2022; Sun et al., 2024; San-Millán et al., 2022). Cardiolipin is a unique phospholipid and is required for the optimal activity of inner mitochondrial membrane proteins, including the enzyme complexes of the electron transport chain and ATP production (Paradies et al., 2019). Cardiolipin also plays a role in mitochondrial membrane morphology, stability, dynamics, and in mitochondrial biogenesis (which primarily occurs during sleep when CO2 levels rise). Therefore, it is no surprise that the effects of chronically elevated lactate levels on mitochondrial function may be why it is so strongly associated with type II diabetes (Crawford et al., 2010).
Otto Warburg won the Nobel Prize in Physiology in 1931 for his discovery of the “Warburg Effect”, referring to the observation that many cancer cells produce large quantities of lactate, with glucose not being completely oxidised (into carbon dioxide; Warburg, 1956). In the cases of these tumours, given that they are predominantly running on glycolysis – not the efficient oxidative phosphorylation process – focusing on restoring effective respiration would be the most sensible step in addressing Warburg Effect-displaying tumours, as well as similar diseases involving degenerative metabolism and inflammation.
Examples in nature
Bats have a very high metabolism, as well as an extremely long lifespan for an animal of their size and weight. While similarly-sized animals tend to live for only a few years, bats can often live for a few decades. One plausible explanation for this has to do with their caves. Bat caves typically have more carbon dioxide than the outside atmosphere, but they also usually contain a large amount of ammonia. However, bats maintain a high level of carbon dioxide in their blood, which protects them from the otherwise toxic effects of the ammonia (ammonia also promotes glycolysis and lactate production). Given that higher altitude is strongly positively correlated with lower mortality (Burtscher, 2014), and so is carbon dioxide saturation with altitude, it wouldn’t be much of a stretch to say that carbon dioxide is having a protective effect on bat lifespan.
Similarly, naked mole rats are another small animal with a very long lifespan. In captivity they live up to 30 years old, and a little shorter in the wild, roughly 9 times longer than similarly-sized mice. They also live underground, in closed burrows with higher carbon dioxide levels. Native to the high-altitude plains of Ethiopia and Kenya, they would naturally have higher carbon dioxide levels in their tissues, perhaps helping explain their long lifespans.
Like mole rats, bees also live in closed, high carbon dioxide environments. Interestingly, in colonies of either organism, there is a queen, and it is her who is the only female that reproduces. The queen bee is the largest individual in the colony, and her workers happen to carefully control the concentration of carbon dioxide, because it must be kept high in order to accelerate ovary action and induce egg laying (Koywiwattrakul et al., 2005). Since queen bees and mole rats live in the dark, their high carbon dioxide levels probably compensate for the lack of light, because both light and CO2 help to maintain oxidative metabolism and inhibit lactate formation.
In humans, it has been observed that higher carbon dioxide levels, at altitude, can protect against obesity and metabolic complications. Obese adults with metabolic syndrome – who could eat freely and were not allowed to exercise – were exposed to an altitude of 2,650 m, and their metabolic rates increased by an average of about 20% and roughly 3 kgs were lost by each subject after only 2 weeks (Lipp et al., 2010). Similarly, Butterfield et al. (1992) observed that adults taken to 4,300 m showed a 27% rise in basal metabolic rate on only the second day of testing. On average, the subjects ate 452 calories more than usual per day, yet still lost over 2.1 kg of adipose tissue after 2 weeks. Interestingly, during strenuous exercise, the subjects were able to expend a whopping 37% more energy than at sea level (Butterfield et al., 1992). This likely explains why elite endurance athletes tend to train at altitude before racing season.
Furthermore, construction workers in India who transferred from low altitude areas to working at 2,750 m in the Himalayas maintained their body weight despite eating between 400-2,900 extra calories per day (Sridharan et al. , 1987).
A 2025 systematic review and meta-analysis concluded that moderate passive hypoxic conditioning, in other words, increasing one’s tolerance to carbon dioxide, could be an effective therapy in managing adipose mass without potential health risks, like those seen in other current therapies (Timon et al., 2025). Figure 1 compares the topography of the United States with geographically distributed obesity rates. Coincidence?
While altitude itself is a complex stimulus and not necessarily equivalent to carbon dioxide therapy, these findings are still consistent with the broader idea that metabolism can be shifted away from the low-energy, lactate-dominant state typical of obesity and related degenerative conditions and towards a more efficient, oxidative state.
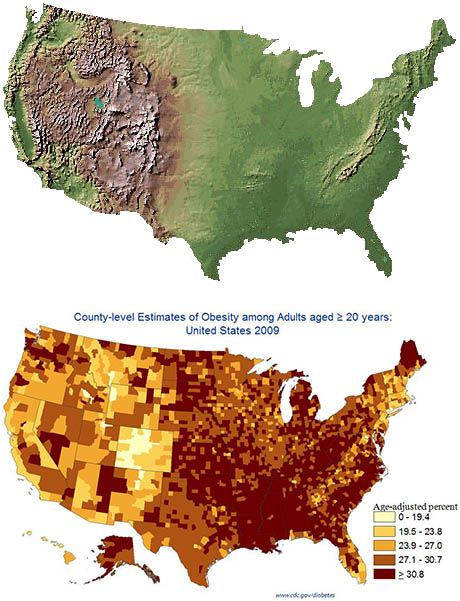
Figure 1. Composite illustration used in blog coverage of Voss et al. (2013), combining a U.S. topographic relief map (USGS National Elevation Dataset) with the CDC’s 2009 county-level age-adjusted adult obesity map.
Conclusion
Taken together, it is clear to see that carbon dioxide’s functions stretch far beyond blood acidity, and I believe it shouldn’t be merely viewed as a waste product of mitochondrial respiration. Carbon dioxide appears to have an evolutionary alignment with our physiology, as well as with those in other organisms, and its numerous actions on fundamental biological processes in a positive and coherent way is what constitutes its broad protective role. Restoring the conditions that allow carbon dioxide to be produced and retained may likely prove to be a fundamental and safe way we can restore metabolic health and protect ourselves from degenerative diseases.
References
- Burtscher, M. (2014). Effects of Living at Higher Altitudes on Mortality: A Narrative Review. aging and disease. doi:https://doi.org/10.14336/ad.2014.0500274
- Butterfield, G.E., Gates, J., Fleming, S., Brooks, G.A., Sutton, J.R. and Reeves, J.T. (1992). Increased energy intake minimizes weight loss in men at high altitude. Journal of Applied Physiology, 72(5), pp.1741–1748. doi:https://doi.org/10.1152/jappl.1992.72.5.1741
- Curley, G., Laffey, J.G. and Kavanagh, B.P. (2010). Bench-to-bedside review: carbon dioxide. Critical care (London, England), [online] 14(2), p.220. doi:https://doi.org/10.1186/cc8926
- Crawford, S.O., Hoogeveen, R.C., Brancati, F.L., Astor, B.C., Ballantyne, C.M., Schmidt, M.I. and Young, J.H. (2010). Association of blood lactate with type 2 diabetes: the Atherosclerosis Risk in Communities Carotid MRI Study. International Journal of Epidemiology, [online] 39(6), pp.1647–1655. doi:https://doi.org/10.1093/ije/dyq126
- Kimura, D. (2006). HYPOCAPNIA- AND HYPERCAPNIA- INDUCED IMMUNOMODULATION IN… : Critical Care Medicine. [online] Available at: https://journals.lww.com/ccmjournal/pages/articleviewer.aspx?year=2006&issue=12002&article=00150&type=Fulltext [Accessed 4 Apr. 2026].
- Kamel, K.S., Oh, M.S. and Halperin, M.L. (2019). L-lactic acidosis: pathophysiology, classification, and causes; emphasis on biochemical and metabolic basis. Kidney International, 97(1). doi:https://doi.org/10.1016/j.kint.2019.08.023
- Koywiwattrakul, P., Thompson, G.J., Sitthipraneed, S., Oldroyd, B.P. and Maleszka, R. (2005). Effects of carbon dioxide narcosis on ovary activation and gene expression in worker honeybees, Apis mellifera. Journal of Insect Science, 5(1). doi:https://doi.org/10.1093/jis/5.1.36
- Lewis, J.L. (2025). Respiratory Alkalosis. [online] MSD Manual Professional Edition. Available at: https://www.msdmanuals.com/professional/nephrology/acid-base-regulation-and-disorders/respiratory-alkalosis
- Lippl, F.J., Neubauer, S., Schipfer, S., Lichter, N., Tufman, A., Otto, B. and Fischer, R. (2010). Hypobaric Hypoxia Causes Body Weight Reduction in Obese Subjects. Obesity, 18(4), pp.675–681. doi:https://doi.org/10.1038/oby.2009.509
- Liu, W., Wang, Y., Bozi, L.H.M., Fischer, P., Jedrychowski, M.P., Xiao, H., Wu, T., Darabedian, N., He, X., Mills, E.L., Burger, N., Shin, S., Reddy, A., Sprenger, H.-G., Tran, N., Winther, S., Hinshaw, S.M., Shen, J., Seo, H.-S. and Song, K. (2023). Lactate regulates cell cycle by remodeling the anaphase promoting complex. Nature. [online] doi:https://doi.org/10.1038/s41586-023-05939-3
- Longhitano, L., Vicario, N., Tibullo, D., Cesarina Giallongo, Broggi, G., Caltabiano, R., Maria, G., Altieri, R., Baghini, M., Michelino Di Rosa, Parenti, R., Giordano, A., Maria Caterina Mione and Giovanni Li Volti (2022). Lactate Induces the Expressions of MCT1 and HCAR1 to Promote Tumor Growth and Progression in Glioblastoma. Frontiers in Oncology, 12. doi:https://doi.org/10.3389/fonc.2022.871798
- Paradies, G., Paradies, V., Ruggiero, F.M. and Petrosillo, G. (2019). Role of Cardiolipin in Mitochondrial Function and Dynamics in Health and Disease: Molecular and Pharmacological Aspects. Cells, 8(7), p.728. doi:https://doi.org/10.3390/cells8070728
- Randle, P.J., Garland, P.B., Hales, C.N. and Newsholme, E.A. (1963). THE GLUCOSE FATTY-ACID CYCLE ITS ROLE IN INSULIN SENSITIVITY AND THE METABOLIC DISTURBANCES OF DIABETES MELLITUS. The Lancet, 281(7285), pp.785–789. doi:https://doi.org/10.1016/s0140-6736(63)91500-9
- Salvati, K.A. and Beenhakker, M.P. (2019). Out of thin air: Hyperventilation-triggered seizures. Brain research, [online] 1703, pp.41–52. doi:https://doi.org/10.1016/j.brainres.2017.12.037
- Sridharan, K., A.K. Mukherjee, S.K. Grover, M.M.L. Kumaria, B.S. Arora, and R.M. Rai 1987. Assessment of nutritional status and physical work capacity of road construction workers at altitude of 2,150–2,750 m on two different ration scales. Nutr. Rep. Int. 35:1269–1277
- Iñigo San-Millán, Sparagna, G.C., Chapman, H.L., Warkins, V.L., Chatfield, K.C., Shuff, S., Martinez, J.L. and Brooks, G.A. (2022). Chronic Lactate Exposure Decreases Mitochondrial Function by Inhibition of Fatty Acid Uptake and Cardiolipin Alterations in Neonatal Rat Cardiomyocytes. Frontiers in Nutrition, [online] 9. doi:https://doi.org/10.3389/fnut.2022.809485
- Staubli, M., Vogel, F., Bartsch, P., Fluckiger, G. and Ziegler, W.H. (1994). Hyperventilation-induced changes of blood cell counts depend on hypocapnia. European Journal of Applied Physiology and Occupational Physiology, 69(5), pp.402–407. doi:https://doi.org/10.1007/bf00865403
- Sun, Z., Ji, Z., Meng, H., He, W., Li, B., Pan, X., Zhou, Y. and Yu, G. (2024). Lactate facilitated mitochondrial fission-derived ROS to promote pulmonary fibrosis via ERK/DRP-1 signaling. Journal of Translational Medicine, [online] 22, p.479. doi:https://doi.org/10.1186/s12967-024-05289-2
- Timon, R., Rojas-Valverde, D. and Camacho-Cardenosa, M. (2025). Effect of passive hypoxia on body fat mass in older and young adults: A systematic review and meta-analysis. Physiology & Behavior, pp.114948–114948. doi:https://doi.org/10.1016/j.physbeh.2025.114948
- Voss JD, Masuoka P, Webber BJ, Scher AI, Atkinson RL. Association of elevation, urbanization and ambient temperature with obesity prevalence in the United States. International Journal of Obesity. 2013;37(10):1407–1412
- Warburg, O. (1956). On the Origin of Cancer Cells. Science, 123(3191), pp.309–314. doi:https://doi.org/10.1126/science.123.3191.309
Leave a comment